
An optical spectrometer is an instrument used to measure properties of light over a specific portion of the electromagnetic spectrum, typically used in spectroscopic analysis to identify materials. A spectroscope helps scientist to study and observe a spectrum of light, This inexpensive but durable handheld spectroscope lets you measure the wavelengths of light over the range of 350 to 750 nm with 5 nm accuracy. What makes this band of light unique is that humans can detect these frequencies with their eyes. The variable measured is most often the light’s intensity but could also, for instance, be the polarization state. Spectrometers are used in many fields. For example, they are used in astronomy to analyze the radiation from astronomical objects and deduce chemical composition. Scientist also use spectroscope to study ultraviolet and infrared spectrums to determine the elements burning in distant stars.
The spectrometer uses a prism or a grating to spread the light from a distant object into a spectrum. Construction eliminates internal reflections to provide the sharpest, clearest spectrum possible. Comes with instructions and an introduction to the nature of light. This allows astronomers to detect many of the chemical elements by their characteristic spectral fingerprints. If the object is glowing by itself, it will show spectral lines caused by the glowing gas itself. These lines are named for the elements which cause them, such as the hydrogen alpha, beta, and gamma lines. Chemical compounds may also be identified by absorption. Typically these are dark bands in specific locations in the spectrum caused by energy being absorbed as light from other objects passes through a gas cloud. Much of our knowledge of the chemical makeup of the universe comes from spectra.When a material is heated to incandescence it emits light that is characteristic of the atomic makeup of the material. Particular light frequencies give rise to sharply defined bands on the scale which can be thought of as fingerprints.
The light passed through a prism that refracted the beam into a spectrum because different wavelengths were refracted different amounts due to dispersion. This image was then viewed through a tube with a scale that was transposed upon the spectral image, enabling its direct measurement.
A spectroscope helps us find out what stars are made of. It disperses, or separates, white light from a star into a very wide spectrum of colors — much wider than a normal prism does. When spread very wide, black lines appear in the spectrum.
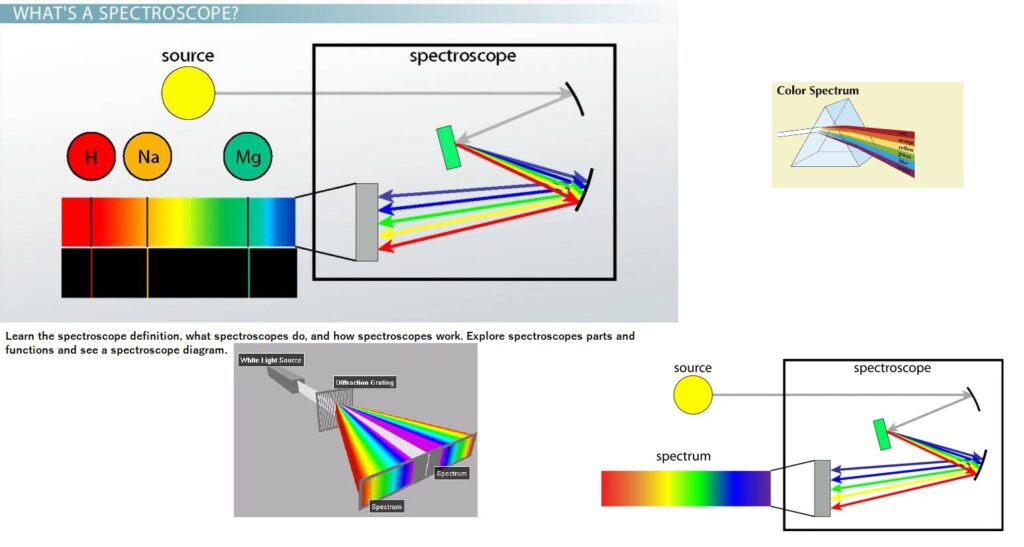

What Is Spectroscopy?
Spectroscopy is the study of the spectra produced when materials interact with or emit light. This is the key to reveal details that cannot be revealed through a picture. A spectrograph – sometimes called a spectroscope or spectrometer – breaks light from a material into its component colors in the same way that a prism splits white light in a rainbow. It records this spectrum, which allows scientists to analyze the light and discover the properties of the materials it interacts with. Spectroscopy is just as important as imaging to understanding the universe.
Absorption spectra of carbon, oxygen, nitrogen and iron. Each element emits a unique pattern of colors, or wavelengths, of visible light. Astronomers can analyze the patterns of light given off by an object like a star to find out what elements it is made of.

How Does a Spectroscope Work?
A spectroscope takes incoming light and shines it through a prism. The prism refracts white light and splits it into different colors. Colored lights bounce off the beams to point in different directions. As a result, the viewer is able to see the colors in a band as absorption lines. Absorption lines are dark lines on a spectrum that can be used to determine what elements are found in an object based on the location of the absorption line. Different colors are different wavelengths of light. A wavelength is the distance between two peaks or two troughs. Wavelength is used to determine the frequency of a light wave and the distance the wave can travel. Light waves with longer wavelengths have a lower frequency and travel longer distances. Also they have less energy. Red, orange, yellow and green colors of light have longer wavelengths. Light waves with shorter wavelengths have higher frequencies and travel shorter distances. They have low energy and are blue, indigo and violet colors of light.
Spectroscopes also serve to help scientists determine the chemical composition of atoms. When atoms are excited, they emit light and this light can be focused through a spectroscope to form an atomic spectrum. An atomic spectrum can be compared to absorption lines in a spectrum. An atomic spectrum is a set of colored lines produced by a spectroscope as a result of the interactions of excited elements. Hydrogen, carbon and oxygen produce spectrum patterns that differ. The absorption lines in the atomic spectrum of a known element and the spectrum of an unknown material can be compared to determine what elements the unknown material is composed of.
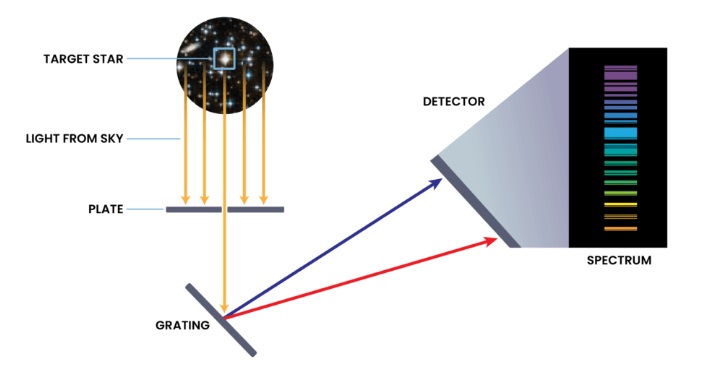
How Do You Read a Spectrum?
Light carries information about the material it interacts with. Different materials interact with light in different ways, and we can use light to understand what something is made of. All matter is made of atoms. Electrons move around the nucleus of an atom at different allowed energies, like rungs on a ladder. Light with the exact energy needed to go between phases can be absorbed, but not others. The electrons fall to the lower rungs, emitting light at specific energies of the gap between the rungs. This allows different atoms and molecules to emit different colors of light. The spectrum of sodium does not look like the spectrum of nitrogen – nor does the spectrum of any other element.
Key Terms
Absorption spectrum – a record of the wavelengths (or frequencies) of electromagnetic radiation absorbed by a substance; The absorption spectrum of each pure substance is unique.
CCD Camera/Array – A charge coupled device that converts an optical signal (light) into an electrical signal for transfer to a video display or computer.
Collimator — An optical element that aligns incoming light rays so that they are parallel.
Diffraction grating – a dispersive element having a surface with very fine, closely spaced grooves, which reflect or refract (bend) different wavelengths of light by different amounts.
Emission spectrum – the colors of the light emitted by the hot gas. An emission spectrum is typically viewed through a slit, and the viewing optics produce an image of the slit in any given color of light. The emission spectrum appears as a line of colored lines, and is thus commonly called spectral lines.
Imaging spectrometer – an imaging instrument capable of determining the radiant intensity of the spectral components of a surface being imaged.
monochromator – a device that selects discrete wavelengths of light; This often includes a diffraction grating.
spectrograph – instrument for dispersing light into its spectrum of wavelengths then for photographing that spectrum.
Spectrometer – an instrument for determining the radiation intensity of atomic spectra.